The Changes in Forest Colors
Fall is an amazing season. The variety of the green shades of spring and summer, now blaze into yellows, oranges, and reds. What is the mechanism that provides us with such colors of the season?
At the risk of entering into the realm of physics and biology, it is necessary to understand something about how sunlight behaves when it reaches the grasses, shrubs, and trees around our homes.
To our senses, light appears invisible. We don’t really see it traveling through the air. Its presence is only detected when it strikes an object. The box is brown, the cup is red, and the sweater is blue. We ascribe those colors to the object, with sunlight (or lamp light) showing the colors there.
In reality, though, sunlight (and lamp light) is made up of all the colors we see in objects. Isaac Newton, the 17th-century scientist, was able to understand and explain how this is true. Puncturing a small hole in the shade of a darkened room and then allowing the ray of sunlight to pass through a glass prism, he was able to demonstrate that sunlight is composed of a range of colors: reds, oranges, yellow, green, blue, indigo, and violet, the “ROYGBIV” acronym we learned in school. Each color corresponds to a specific wavelength of energy. The red wavelengths are longer than the indigo and violet wavelengths.
So then, how is it we see colors? The object’s color is the result of chemicals on the object’s surface that absorb and reflect particular wavelengths of light. Here’s the tricky part, if an object is, let’s say red, the object is really absorbing not the red light wavelengths, but everything except red. The red wavelengths are reflected from the object and that red light is what makes it to our eyes. The object is ‘grabbing on to’ the blue, yellow, green, violet light wavelengths!
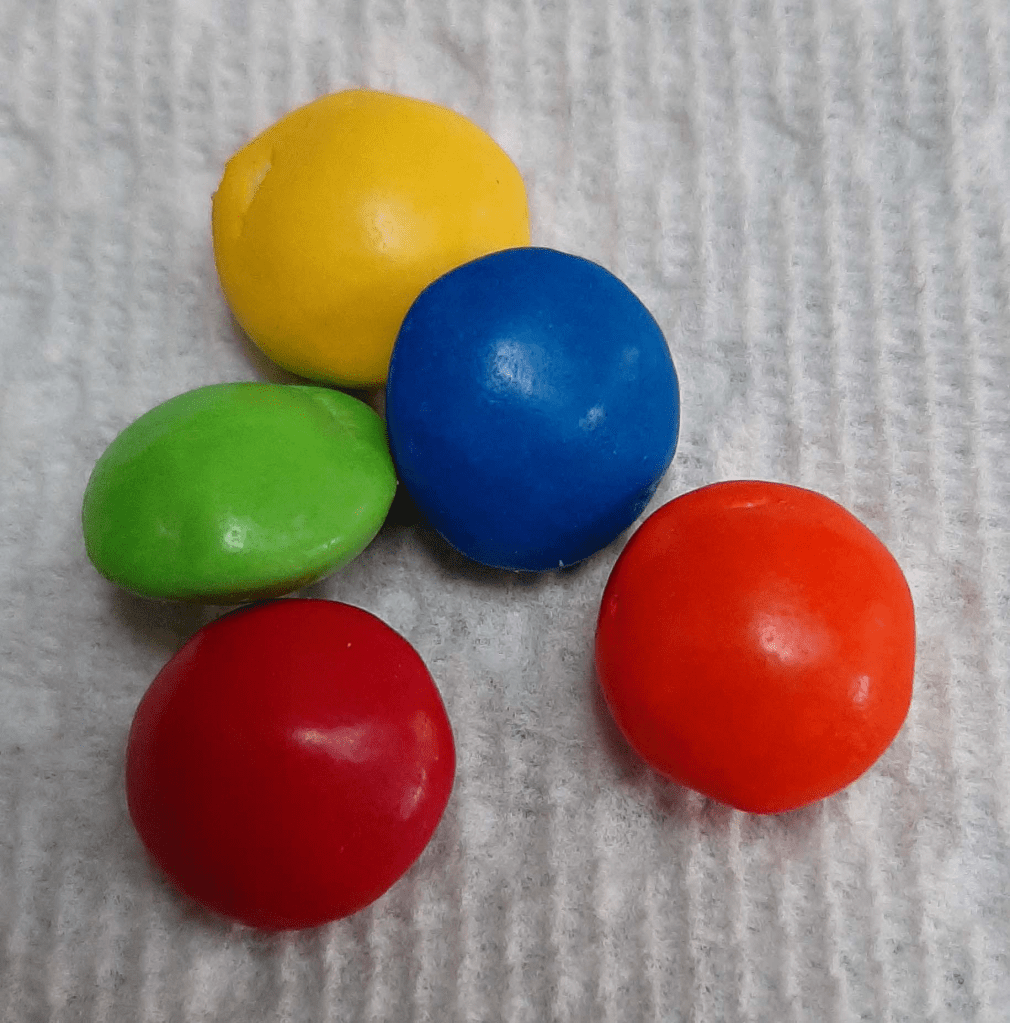
Take a look at the pieces of candy above. The colors of the outer cover are the result of light energy being absorbed and reflected by surface pigments. The yellow dye of the top candy piece absorbs reds, blues, and greens, but the yellow light is reflected off the candy. The same process is true for each of the other colors. The color of the candy is that wavelength being reflected back to our eyes.
Here’s a question to ponder: What happens to the various light wavelengths when an object is white, like the paper this is printed on?
OK, so, what does all this have to do with forest colors?
Remember I said something at the start of this essay about the various shades of green that we see in plants during the spring and summer? We know now, that the leaves and stems of the plants are absorbing the variety of wavelengths except green. The shades result from a mixing of the pigments in the plant structures that allow for a blending of color wavelengtare. The color wavelength energies that the plant absorbs is what the plant uses to perform photosynthesis, the combining of carbon dioxide (CO2) and water to produce sugars and oxygen. Green energy wavelengths are not absorbed, but reflected back into the environment. However, there must be pigments in the leaves that absorb those non-green wavelengths.
All spring and summer, plants combine water and CO,2 with solar energy making the tissues needed to produce leaves, stems, roots, flowers, and eventually, seeds as well as producing the energy needed to run all the process that keeps the plant alive.
What happens when the length of daylight lessens, the availability of water decreases, and the daytime temperatures decrease? These are the conditions the plants must face as the summer season ends and fall starts.
At this time of year here in New England, plants are in the process of ‘shutting down’ photosynthesis. They are getting ready for a time of hibernation. Any light available during the day becomes less available. Daily colder temperatures will slow chemical reactions. Soon any ground water near plant roots will freeze along with any water in tender leaves and stems. If these ice crystals grow large, they will damage soft tissues. The mechanism then in play is for the plants to pull water and sap down into their roots. Tender leaves will be allowed to wither and drop off from stems. This strategy also protects tree branches from accumulating heavy snow loads during winter. The snow loads could result in branches breaking from the tree trunk.

With photosynthesis slowing and then eventually stopping, the green pigment chlorophyll is destroyed. The other pigments, masked all summer long, now become apparent. The orange, yellow, and brown colors from the carotenoid pigments, found in corn, carrots, and bananas along with anthocyanin pigments that give color to apples, Concord grapes, blueberries, and plums give color to the forest trees.
The fallen forest leaves do provide a service to the forest ecosystem. The yearly leaf deposit allows for the forest floor to build up nutrients as the leaves decompose. The decomposed forest soil sits waiting for next year’s spring, ready to release the nutrients made in leaves from years past back to the trees.
